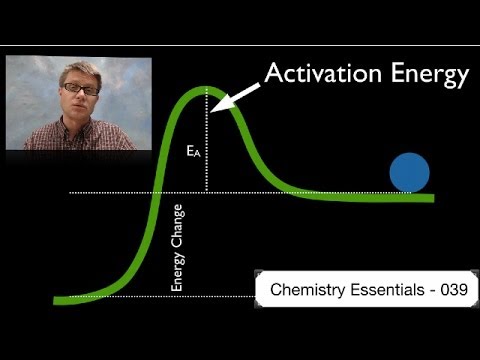
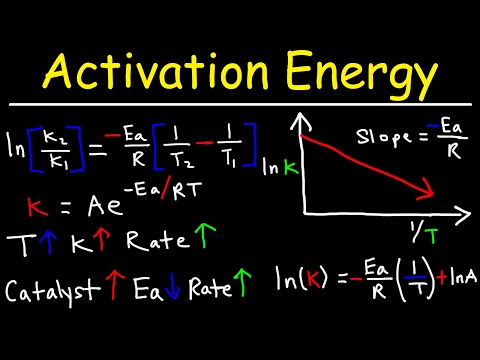
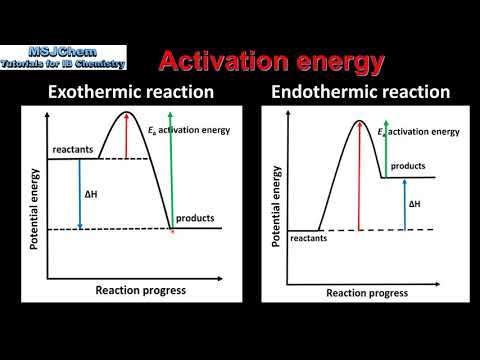
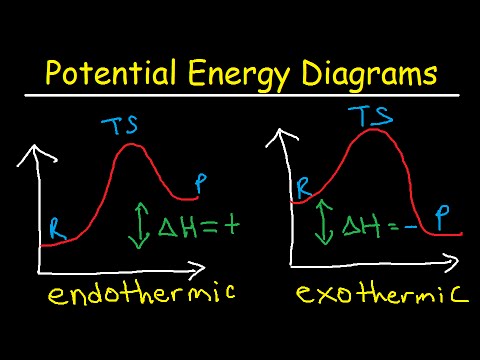
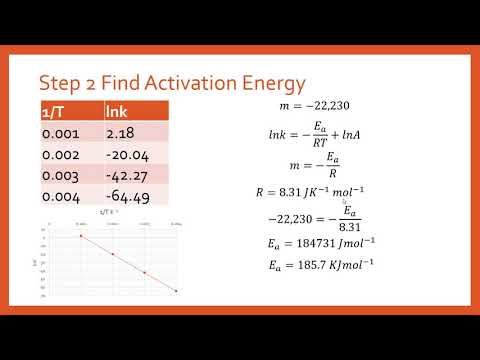
関連ワード:
how do you find the activation energy of a reaction how do you find the activation energy of a reverse reaction how do you calculate the activation energy of a reaction how do you lower the activation energy of a reaction how to find the activation energy of a reaction how the activation energy of a reaction how do enzymes lower the activation energy of a reaction how do enzymes affect the activation energy of a reaction how do enzymes reduce the activation energy of a reaction how do enzymes lower the activation energy of a reaction quizlet